Laboratory tests conducted by Zoono Group (ASX: ZNO) on its Z-71 Microbe Shield surface sanitiser have confirmed the product to be more than 99.99% effective against feline coronavirus, a surrogate of COVID-19.
While the virus has been shown to survive on surfaces for up to nine days, Z-71 was successfully tested against a variety of pathogens for up to 30 days on surfaces and 24 hours on hands.
Z-71 was previously tested in 2014 against bovine coronavirus; however, this latest strain has required the product to be re-tested.
Two separate tests were completed to EN Standard 14476:2013+A2:2019 for chemical disinfectants and antiseptics.
It is the European standard which applies to products within the medical area including hygienic hand rubs, hygienic hand wash, instrument disinfection by immersion, surface disinfection by wiping, spraying, flooding or other means.
The first was against Vaccinia – sometimes referred to as the ‘mother ship’ of double enveloped viruses which are particularly hard to inactivate.
The results confirmed efficacy of more than 99.99% for Zoono Z-71 Microbe Shield, which incorporates the same technology used in Zoono hand sanitiser.
The subsequent test was against the nominated and globally-accepted surrogate for COVID-19 known as feline coronavirus.
It also confirmed efficacy of greater than 99.99%.
Zoono said the results further demonstrate the ability of its technology to be part of the solution to prevent and protect against the spread of the COVID-19 virus.
Share price rise
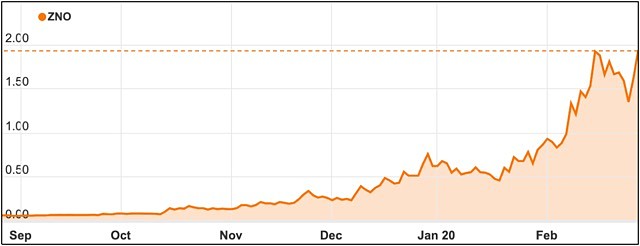
Zoono shares have soared recently as its continues to successfully test its products against coronavirus.
The company’s stock has tripled in price in the last month, following a trend which first appeared in December before the global outbreak was announced.
This week, managing director and chief executive officer Paul Hyslop sold a 10% stake in the company for more than $23 million.
Mr Hyslop recently spoke with Small Caps about the company’s market success over the past few months.
