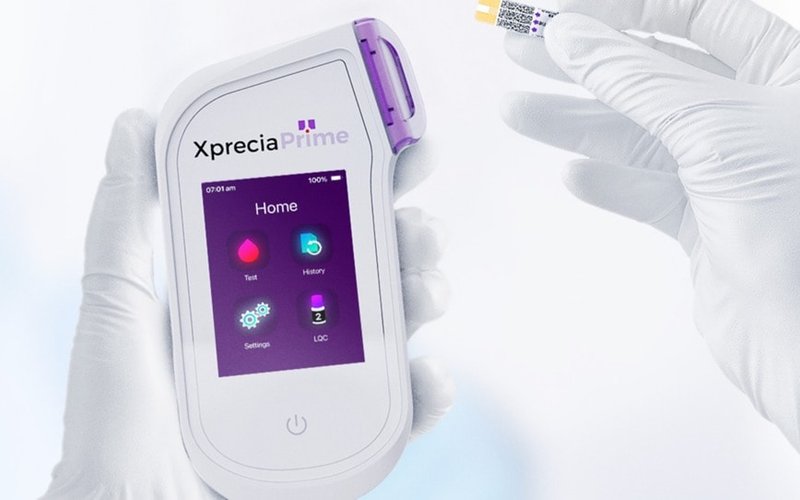
Melbourne-based medical company Universal Biosensors (ASX: UBI) has received two approvals from the US Food and Drug Administration (FDA) for its Xprecia Prime coagulation analyser as a class II device.
Receipt of FDA 510(k) and a clinical laboratory improvement amendments (CLIA) waiver means the company is now poised to sell the product to professional health care clients such as hospitals, clinics and medical practices in the US.
Xprecia Prime is a portable unit designed for fast and reliable prothrombin time and international normalised ratio (PT/INR) testing, allowing doctors to monitor vitamin K antagonist oral anticoagulation therapies such as Warfarin and Coumadin at the point of care.
Dangerous bleeding events can occur if the ideal dosage of a blood-thinning drug is exceeded and patients can be at a risk of thrombosis if the dosage is lower than required.
‘Historic moment’
Chief executive officer John Sharman said the approval was an “historic moment” for the company.
“This represents more than ten years of research and development work and many millions of dollars of investment,” he said.
“This is the first time the FDA has granted a CLIA waiver by application to any coagulation device and it is testament to the performance of Xprecia Prime.”
Unrestricted access
The number of PT/INR tests performed in clinics is believed to be the largest part of the US market, with more than 140 million test strips sold each year and more than six million residents taking blood-thinning medication.
“To have won unrestricted access to all clinics and hospitals across the US is a major achievement,” Mr Sharman said.
“This FDA approval represents the first opportunity in Universal’s history to access the lucrative and fully-reimbursed US market [and] we expect Xprecia Prime will qualify under the existing reimbursement codes used by Medicare, Medicaid and USA health insurers.”
Biosensor development
Universal is developing and commercialising a range of biosensors in oenology (the wine industry), human health (including oncology, coagulation, women’s health and fertility) and environmental testing using patented platform technology and hand-held point-of-use devices.
With the latest approvals milestone, Mr Sharman said the company could now look forward to finalising a pipeline of sales and distribution contracts to help “win new market share and generate substantial revenues”.
Get the wire before the market opens.
The ASX small-cap stories that matter, filed before 9am AEST. Curated by the Small Caps desk.