On the back of promising proof-of-concept results on a range of cancers, Invion Group (ASX: IVX) is looking to advance its novel photodynamic therapy (PDT) technology for the treatment of atherosclerosis and infectious diseases, and expand into new markets.
The life sciences company is focused on developing Photosoft technology as an improved next-generation PDT, which uses non-toxic photosensitisers and visible light in combination to produce cytotoxic-reactive oxygen that kills malignant cells, shuts down tumours and potentially stimulates the immune system.
It is considered a potential alternative to surgery or the immunosuppressive treatment approaches of radiotherapy and chemotherapy.
Invion holds Photosoft license rights in Australia and New Zealand with the technology licensor RMW Cho Group funding research and clinical trials via a research and development partnership.
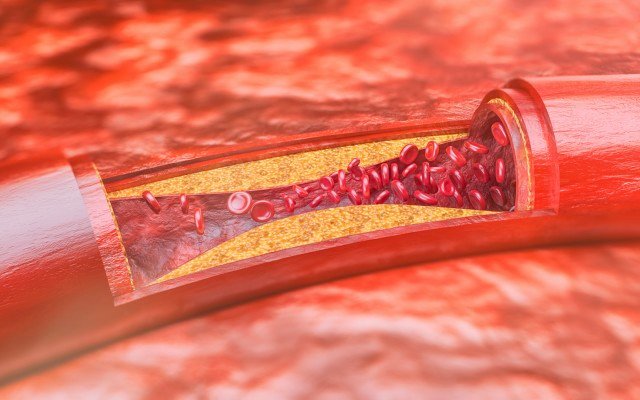
The pair also recently inked a deal for Invion to acquire a license and exclusive distribution rights to use the technology on atherosclerosis, which is the build-up of fats and cholesterol in and on your artery walls, and infectious diseases including coronaviruses for the Asia Pacific region (excluding Greater China).
There are very significant market opportunities for the two diseases. Atherosclerosis is a key contributor to heart disease – the leading cause of death in the world.
Meanwhile, the COVID-19 pandemic has put the spotlight on new treatment options for infectious diseases.
Proof of concept results
Invion excited the market in late May with promising results from proof-of-concept tests on its latest active pharmaceutical ingredient (API), INV043.
In both in vitro and in vivo studies undertaken by the world renown Hudson Institute of Medical Research, INV043 demonstrated very strong activity against a range of solid tumour types including “significant regression observed in vivo in T-cell lymphoma, triple negative breast and pancreatic cancer models”.
Performance was more than 50 times better than Invion’s previous API, INV-P03, and studies showed INV043 has more than 600 times greater phototoxicity than the widely used photosensitiser, Talaporfin sodium.
What’s more, INV043 was observed to only affect cancer cells and not healthy tissue.
Results also indicated INV043’s potential to be used as a diagnostic tool as the API localises into the tumour and causes it to “glow” under blue light.
“Fluorescence was readily visible even in metastatic deposits as small as less than 1mm, which would otherwise be invisible to the naked eye,” Invion reported.
At the time, Invion chairman and chief executive officer Thian Chew said these initial proof-of-concept results demonstrated the potential of Photosoft technology’s applications in cancer treatment.
“Our next steps include performing further proof-of-concept studies looking at INV043’s effect on the immune response as well as exploring its potential to work together with other therapies,” he added.
Group head of the Ovarian Cancer Biomarkers Research Group at Hudson, Dr Andrew Stephens, called these early indications “promising” and said they “may lead to new treatment options for some of the most difficult to treat cancers”.
Asia Pacific license and distribution deal
Shortly after revealing the test results, Invion announced it had entered into an exclusive distribution and licencing deal with RMW Cho Group to co-develop the next generation PDT technology for the treatment of atherosclerosis and infectious diseases.
Atherosclerosis is a disease in which plaque builds up inside arteries and is regarded a major contributor to cardiovascular diseases. The global market for atherosclerosis treatments is forecast to hit US$56.6 billion (A$79 billion) by 2027 and Asia Pacific is believed to be the fastest growing market.
Under the agreements, Invion will gain exclusive distribution rights to the Photosoft technology in Asia Pacific for these indications.
Invion will pay RMW a one-time amount of $2.25 million as its contribution to the Photosoft development costs.
As part of the transaction, Invion will raise $4.5 million via a share placement priced at $0.014 per new share. This issue price represented a 2.1% discount to the five-day volume weighted average price as at 2 June 2021.
Invion will seek shareholder approval for this co-development agreement and proposed placement at a general meeting scheduled for 23 September.
Financial position
Invion is a very unique ASX biotech as its cancer treatment development program is fully funded by RMW Cho through a research and development services agreement. This means Invion did not have to raise capital to advance its cancer studies.
The company held more than $1 million in cash reserves at the end of the June quarter and changes in the cash position from quarter to quarter are influenced by the timing of payments and receipts.
Invion invoices RMW Cho on a monthly basis and Invion believes it is adequately funded to advance its current program.