Clinical-stage pharmaceutical company Incannex Healthcare (ASX: IHL) has achieved another milestone with the development approval for new product targeting the $15 billion sleep apnoea market.
An Investigational New Drug (IND) application has now been successfully submitted to the US Food and Drug Administration (FDA) for its unique IHL-42X development, a product it believes can relieve many of the issues related to obstructive sleep apnoea treatment.
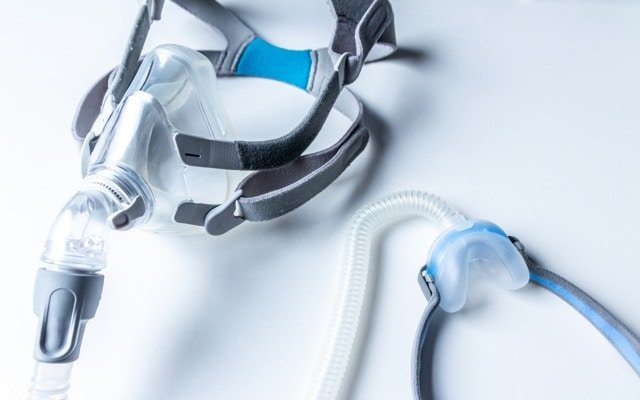
Currently the leading sleep apnoea treatment is continuous positive airway pressure (CPAP) therapy. For many, the use of CPAP machines at night is too uncomfortable.
CPAP involves strapping a mask on to the persons face and that mask is connected to a hose and an air pump that forces pressurised air into the persons nose and mouth while they attempt to sleep.
Much simpler treatment
Incannex’ IHL-42X treatment uses a dose made up of a combination of dronabinol, a synthetic form of tetrahydrocannabinol (THC), and acetazolamide, a carbonic anhydrase inhibitor.
Earlier results from a Phase 2 proof of concept clinical trial found that IHL-42X reduced average apnoea-hypopnoea index (AHI) by a median of 50.7% versus baseline assessments and 25% of participants experienced greater than an 80% reduction in the AHI.
Importantly, no serious treatment adverse events were reported during the clinical trial and THC levels in the blood were found to be below the limits for impaired driving the morning after nocturnal dose administration.
Next set of trials
Incannex has now submitted an IND dossier to the FDA which is made up of comprehensive modules on the safety and efficacy of IHL-42X and its component active pharmaceutical ingredients.
It also includes detailed information on the development, manufacturing, quality and stability of the IHL-42X drug product, as well as the clinical protocol and investigator information for the Phase 2/3 IND opening clinical trial.
Incannex managing director and chief executive officer, Joel Latham, said the submitting of an IND to the FDA is crucial for companies to gain regulatory approval, conduct clinical trials, and engage in scientific dialogue with FDA whilst they progress investigational drugs through the stages of development in the US.
“The FDA review process for an IND application involves evaluation of the scientific, clinical, and safety aspects to ensure that the proposed clinical trial meets regulatory requirements.”
The company’s IND submission includes details of the clinical trial protocol for the pivotal IND opening clinical trial.
This will be a multi-site phase 2/3 clinical trial which will assess IHL-42X at the best performing two doses from the successful phase 2 proof- of-concept clinical trial, in patients with OSA who are intolerant, non-compliant, or naïve to positive airway pressure, over a 52-week treatment period.
Planning for the trial includes the dosing of participants with one IHL-42X, dronabinol, acetazolamide or a placebo for the entirety of the trial.
The participants will all complete daily surveys on their sleep quality, attend monthly clinic visits to assess functional outcomes of sleep, cognitive function and other measures of safety and efficacy.
At the next stage overnight polysomnography will be conducted every three months, to determine the effect of treatment on the patients’ Apneoa Hypopnea Index score (AHI) along with a range of other sleep parameters. All drug treatments will be compared to placebo.
Principal trial investigators named
Incannex announced in June that Dr John Douglas Hudson, board certified in neurology and sleep medicine and Dr Russell Rosenberg who holds a doctorate in clinical and research psychology from The Ohio State University will act as the principal investigators for the trials.