Just over a month since its burns treatment system was approved for FDA’s compassionate use Investigational Device Exemption (IDE) program, regenerative medicine company AVITA Medical (ASX: AVH) has received a further boost courtesy of the US regulator.
The FDA approved AVITA’s Premarket Approval (PMA) application to market the RECELL Autologous Cell Harvesting Device (RECELL System) to treat severe thermal burns in patients 18 years and older.
With the FDA’s approval now in hand, AVITA is moving to finalise its US launch by the end of this year.
The RECELL System is a medical device designed to facilitate skin regeneration while minimising the amount of skin that must be “harvested” at the time of surgery.
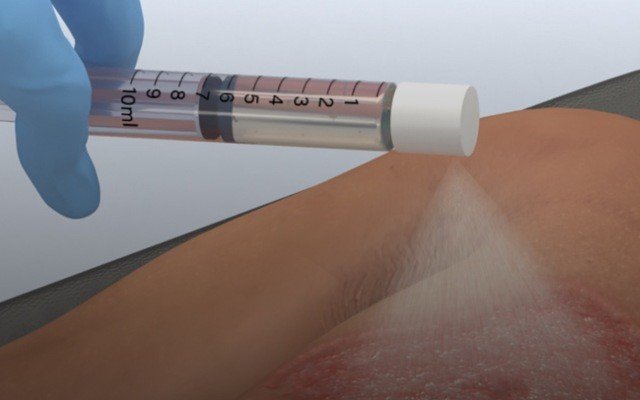
RECELL uses a small amount of a patient’s own skin to prepare “Spray-On Skin Cells” at the point of care in as little as 30 minutes which could potentially revolutionise how severe thermal burns patients are treated in future.
To support its application with the FDA, AVITA conducted two randomised, controlled clinical trials which demonstrated that treatment of acute burn wounds with the RECELL System requiring substantially less donor skin than required with conventional split-thickness autografts to achieve closure of burn wounds.
The first controlled trial compared treatment with the RECELL System versus treatment with conventional split-thickness autografts in patients with second-degree burn injuries.
During the pivotal trial, the patient-donor skin required to be harvested to treat burn sites with the RECELL System was 97.5% less than the amount harvested to treat burn sites using standard care – resulting in a statistically significant reduction in patient-reported pain, increased patient satisfaction and improved donor scar outcomes.
The second randomised, controlled pivotal clinical trial evaluated treatment of third-degree burns with the RECELL System combined with split-thickness autografts.
At eight weeks post-treatment, 92% of the burn sites treated with the RECELL System achieved “complete healing” compared to 85% for the sites treated with standard care, thereby demonstrating “non-inferiority”, a significant milestone that helps to validate the RECELL system.
Although AVITA’s technology is still undergoing practical trials, the effectiveness of its RECELL system is on course to reduce donor skin requirements and provide “key clinical benefits to patients and significant reductions in the cost of treatment,” according to AVITA.
“Patients are at the centre of everything we do at AVITA Medical and we are pleased to be able to provide an innovative solution for the treatment of severe burn injuries,” said Dr Michael Perry, CEO of AVITA Medical.
“Today’s approval of the RECELL System marks an important milestone for us and provides a new way to treat burns for the thousands of patients with significant unmet medical needs. We are grateful to those patients who participated in clinical trials and to the clinical trial investigator teams whose dedication and scientific rigour made this approval possible.”
“We also greatly appreciate our collaboration with BARDA and the support that they have provided to us throughout the development of the RECELL System,” said Mr Perry.
Looking deeper into RECELL
The RECELL system requires a small skin sample from the patient which is then immersed in the company’s proprietary enzyme solution which effectively separates skins cells to produce what AVITA calls “spray-on skin cells”.
The resultant Regenerative Epidermal Suspension (RES) includes keratinocytes, fibroblasts, and melanocytes, which play a critical role in wound healing. The suspension is then sprayed directly onto the prepared burn wound, providing a broad and even distribution of live cells across the entire area that’s been damaged by thermal burns.
According to AVITA, the RECELL System can be used to prepare enough RES to treat a wound up to 80 times the size of the donor skin sample. In practical terms, AVITA says that a skin sample approximately the size of a credit card can be used to treat a wound that covers a patient’s entire back.
A strong market for burns treatment
The frequency of burn-related injuries and the cost of treatment is currently very high, thereby prohibiting wider implementation for more patients.
The Centers for Disease Control and Prevention (CDC) in the US reports that 486,000 patients receive emergency medical treatment for burns annually. Furthermore, burn injuries result in approximately 3,400 deaths each year, the third-leading cause of accidental home injury deaths in the US.
Burns covering up to 90% of a person’s body surface area – once considered fatal injuries –have become survivable with appropriate treatment. Although split-thickness autografts are the current standard treatment, grafting is associated with significant donor site pain, delayed healing and scarring.
AVITA intends to improve upon the outcomes currently being achieved and hopes to capture a significant chunk of its target market for burns, currently estimated to be around US$200 million per year.
Following its recent FDA approval, AVITA Medical said it has immediately undertaken substantial efforts to prepare for its US market launch.
To be able to launch its product in the US, AVITA has set about recruiting a dedicated “field sales team”, conducting direct market research, establishing pricing and reimbursement strategies as well as support infrastructure.
“While severe thermal burns happen every day, in a national security emergency an overwhelming number of people may need burn care quickly,” said Dr Rick Bright, Director of BARDA.
“Medical providers need easy-to-use treatments on hand to save more lives. Our goal is not only to support product development but also to integrate those products into routine care to build preparedness,” said Mr Bright.
This morning’s news saw AVITA Medical’s shares rise 11% to $0.10 a share.